No.2273
Generic Drugs Market in Japan: Key Research Findings 2019
Market Share of Generic Drugs in Volume Approaching Goal of 80%, but Market Environment Becoming Difficult Furthermore
Yano Research Institute (the President, Takashi Mizukoshi) conducted a survey on the domestic generic drugs market, and has found out the trends of market players and future perspectives.
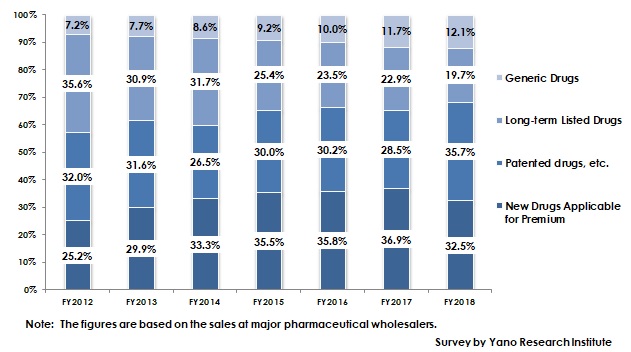
Summary of Research Findings
Generic drugs in Japan has begun being widely accepted after propelled use of generic drugs and improved environment for facilitating the use of the drugs, which were a part of the medical fee restriction measures taken since around 2002.
The ratio of generic drugs in the entire ethical drugs market has been rapidly increasing since around 2013 when the government set the goal to occupy more than 60% in volume. According to Ministry of Health, Labour and Welfare (MHLW), the market share of generic drugs in volume as of September 2018 rose by 6.8 points from the previous year to attain 72.6%, approaching the goal of reaching 80% by September 2020. On the other hand, however, the market share of generic drugs in value has not been disclosed. From the estimation of the market share based on the sales value at major drug wholesalers was 12.1% as of FY2018. Therefore, the ratio of generic drugs approximates to 10% of the entire ethical drugs market.
The environment surrounding generic drug makers are likely to be difficult more than ever, because patent expiration of major small-molecule drugs have become overall done with, and because generic drugs are likely to face fierce price competition, being chosen by those facilities facing ever-more difficult business operations such as medical institutions, dispensing pharmacies, and pharmaceutical wholesalers.
Though the government policy to encourage the use of generic drugs has been clear, as can see from formulary (the guide to use the most effective and economic medicines for patients at medical institutions) attracting attention, the future of the generic drug industry is still in the maze.
Noteworthy Topics
Is Formulary Effective in Expansion of Generic Drugs Market?
Formulary has suddenly begun being introduced in Japan to follow suit of other countries. Formulary is the guide to use the most effective and economic medicines for patients at medical institutions, which began in Europe and the U.S. in the 1990s.
Because formulary encourages the use of more inexpensive drugs among the drugs with the same kind and same efficacy if no large difference in effect and safety, if the patent of the first chosen drug is expired, generic drugs are to be chosen. Therefore, formulary has begun being paid attention as the next move for encouraging using of generic drugs and suppressing medical fee. More attempts going on, shown by some researches on the status of making formulary at hospitals which is reported in the special survey conducted in 2018 on “the result validation of Revision of the Medical Payment System FY2017”.
Just like St. Marianna University School of Medicine, one of the most famous cases of the domestic introduction, formulary has primarily been introduced to several university hospitals, including Showa University Hospital. In addition to this, Japan Sea Health Care Network, a corporation to propel local medical partnerships, has been reported to start making attempts for creating formulary, which is expected to expand using of formulary at increasing number of corporations propelling local medical partnerships.
Research Outline
2.Research Object: Pharmaceutical manufacturers and wholesalers, medical organizations, dispensing pharmacies, and organizations pertaining to the industry
3.Research Methogology: Face-to-face interviews by the expert researchers, and literature research
The Generic Drugs Market
A generic drug in this research refers to a pharmaceutical drug that contains the same active ingredients with the same amount and the same administration route as a drug that was originally developed. In principle, it has the same efficacy/effects, application and dosage, and is supposed to have the same clinical effects as the original one.
<Products and Services in the Market>
Generic drugs, authorized generic, biosimilar
Published Report
Contact Us
The copyright and all other rights pertaining to this report belong to Yano Research Institute.
Please contact our PR team when quoting the report contents for the purpose other than media coverage.
Depending on the purpose of using our report, we may ask you to present your sentences for confirmation beforehand.