No.2376
Neuromodulation Device Market in Japan: Key Research Findings 2019
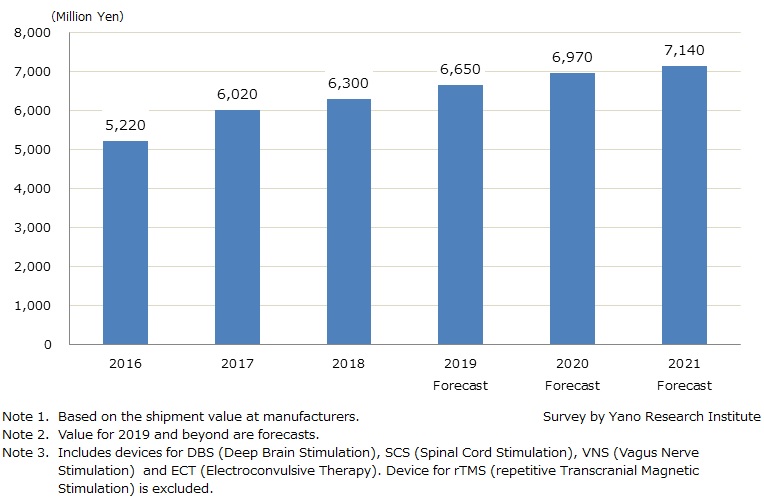
Neuromodulation Device Market for 2018 Yielded 6,300 Million Yen, 104.7% YoY: Deeper Understanding Obtained for Treatments, Rise in Introduction of Devices
Yano Research Institute (the President, Takashi Mizukoshi) has conducted a survey on the neuromodulation device market in Japan, and found out the market size, the trends by device for each therapy, the trends by market players, and the future perspectives.
Market Overview
Neuromodulation treatment is a therapy that uses devices that delivers electric/magnetic stimulation or drugs to control neural activities; as a medical treatment for diseases or disorders, mild electrical stimulation is delivered via devices, or medication is given continually to restore/control functions of central nervous system. Neuromodulation devices are classified broadly into two categories: Invasive device that requires surgery, and noninvasive device.
Examples of invasive devices are Vagus Nerve Stimulation (VNS) devices for refractory epilepsy, Spinal Cord Stimulation (SCS) devices for intractable chronic pain, and Deep Brain Stimulation (DBS) devices for Parkinson’s disease and essential tremors, which are already covered by insurance domestically. As to noninvasive treatments, devices for Electroconvulsive Therapy (ECT) are also covered by insurance domestically, and the treatment is conducted on everyday clinical practice on symptoms of depression, bipolar disorder, and schizophrenia. Moreover, as devices for rTMS (repetitive Transcranial Magnetic Stimulation) obtained pharmaceutical approval in September 2017, it was also listed for insurance coverage in June 2019; it is gathering attention as a new therapy for a type of depression that has no effective drug therapy.
The domestic market of neuromodulation devices for 2018 (based on the shipment value at manufacturers) is estimated to be 6,300 million yen, 104.7% of the preceding year. As neuromodulation treatments obtain acceptance and device installation expands, the market size is projected to increase to 7,140 million yen by 2021.
Noteworthy Topics
Expansion of Insurance Coverage, Terms and Conditions
Insurance coverage has expanded the range of neuromodulation therapies; however, in some areas, restrictive conditions on target patients and facilities are slowing down the speed of penetration.
Regarding the ECT (Electroconvulsive Therapy), it has pervaded as a standard treatment in psychiatry domain for its high therapeutic efficacy. Meanwhile, because of difficulty in staffing of anesthesiologists, the therapy can only be done at general hospitals or psychiatric hospitals with well-equipped facilities. Considering the small number of hospitals with ECT devices today, new installations cannot be expected much in this market.
Similar challenge is noted for rTMS (repetitive Transcranial Magnetic Stimulation), a therapy that is newly covered by insurance. An immediate issue for rTMS is how to spread the treatment domestically after being covered by insurance. While rTMS particularly requires compliance for proper use, i.e. safe and appropriate operations, it should be noted that range of therapy covered by insurance is still very limited.
Future Outlook
The domestic market of neuromodulation devices is expected to make an upward turn hereafter. Factors of growth are such as: (1) penetration/generalization of treatments covered by insurance, (2) advancements of R&D in new application developments, and (3) accumulation and utilization of new knowledge on cranial nerves domain.
For (1), as seen in the case of rTMS (repetitive Transcranial Magnetic Stimulation) devices obtaining pharmaceutical approval as new treatment for depression, being authorized by regulator as a new medical treatment method gives a great advantage to neuromodulation therapy over existing treatments including drugs, surgeries, and cognitive behavioral therapy (CBT). If accumulation and verification on evidence proceeds, terms and limitations for target patients and facilities may be softened. Since there are high needs for neuromodulation as continuous treatment and home treatment, further research and development by academic institutions and businesses are expected.
Regarding (2) and (3), considering advancements in development of new therapies and identification on biomarkers, domain of neuromodulation is anticipating full-scale innovation hereafter. Although challenges like cost of clinical research and difficulties in clarifying action mechanism remain, the field is promising and active in R&D; further developments on new treatment methods are awaited.
While domain of neuromodulation is approached from medical perspective, the market also has many issues such as shady diagnosis and use of devices, and provision of commercial-based treatments. To succeed in sales and businesses in the domestic neuromodulation device market, it is assumed that most important point is to promote R&D or product launch based on evidence.
Research Outline
2.Research Object: Manufacturers of neuromodulation-related devices, related academic societies, etc.
3.Research Methogology: Face-to-face interviews by the expert researchers, surveys by telephone and email, and literature research.
Neuromodulation Device Market in Japan
Neuromodulation is a medical therapy using devices that deliver electric/magnetic stimulation or drugs to control neural activities; as a medical treatment for diseases or disorders, mild electrical stimulation is delivered via devices, or medication is given continually to restore/control functions of central nervous system.
The neuromodulation device market in this research refers to devices for DBS (Deep Brain Stimulation) therapy, SCS (Spinal Cord Stimulation) therapy, VNS (Vagus Nerve Stimulation) therapy and ECT (Electroconvulsive Therapy). The market size is calculated based on the shipment value at manufacturers. Devices for rTMS (repetitive Transcranial Magnetic Stimulation) therapy are not included in the market size.
<Products and Services in the Market>
Devices for following therapies: DBS (Deep Brain Stimulation), SCS (Spinal Cord Stimulation), VNS (Vagus Nerve Stimulation), and ECT (Electroconvulsive Therapy).
Published Report
Contact Us
The copyright and all other rights pertaining to this report belong to Yano Research Institute.
Please contact our PR team when quoting the report contents for the purpose other than media coverage.
Depending on the purpose of using our report, we may ask you to present your sentences for confirmation beforehand.